Medical Isotopes: Another Asset of Nuclear Engineering
Something that all engineers have in common is the desire to tackle real-world challenges. For many nuclear engineers the challenges may be related to the power grid, new reactors, simulations, policy, military, or safeguards, just to name a few. Yet, embedded in nuclear science and engineering is another challenge that's particularly unique because there's a good chance someone you know has directly benefited from it-a branch of nuclear medicine called medical isotopes.
Medical isotopes are a class of radioactive isotopes used by hospitals to monitor the body's functions and destroy cancerous tissues. They're different from x-rays and CT scans that use accelerators to generate radiation. The sharp, penetrating, crystal-clear radiation emitted by certain isotopes allows medical doctors to identify precisely where the isotope has been administered by the body using special cameras-like a biological tracking device. For example, they can be used to monitor blood flow and identify the component of the heart that may be damaged after a heart attack (single-photon emission computed tomography). They can be tagged onto a sugar molecule and diffused into the brain to detect early Alzheimer's (positron emission tomography). Or, they can help destroy a tumor with short-range ionizing particles-leaving the surrounding healthy tissues unharmed (radioisotope therapy). When effective they can reduce the need for surgery or chemotherapy, though these lines of attack are often synergistic.
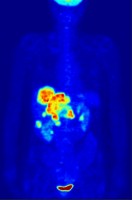 As of 2012 nearly 40 million people every year receive a diagnostic procedure with a medical isotope in what's estimated to be a $5 billion/year industry, according to world-nuclear.org. Radiation therapy extends to the treatment of non-Hodgkins lymphoma, HIV, liver cancer, thyroid cancer, breast cancer, and more. And according to the National Isotope Development Center (isotopes.gov), some of these isotopes are produced through various production routes in a collaboration between universities and Department of Energy-national laboratories.
As of 2012 nearly 40 million people every year receive a diagnostic procedure with a medical isotope in what's estimated to be a $5 billion/year industry, according to world-nuclear.org. Radiation therapy extends to the treatment of non-Hodgkins lymphoma, HIV, liver cancer, thyroid cancer, breast cancer, and more. And according to the National Isotope Development Center (isotopes.gov), some of these isotopes are produced through various production routes in a collaboration between universities and Department of Energy-national laboratories.
Medical isotopes such as Tc-99m, Ac-225, Y-90, F-18, Cu-67, and Sc-47 can be produced by accelerators, high-flux reactors, or harvested from spent fuels and stocks. They need to be purified (quickly) from the initial starting materials and then quickly transported to a collaborating hospital where they can properly labeled (again ... quickly) depending on the patient and the case. Often times an isotope's availability is the Achilles heel of the whole procedure. In 2010 during an actinide conference, I recall a presenter who proudly announced that his team had completely eradicated HIV in a rat using alpha therapy, leaving the rat healthy afterward. The team was ready for the next set of larger clinical trials using chimpanzees, except for one major problem: the team didn't have enough of the isotope to go forward.
Nuclear engineering couldn't be more appropriate for medical isotopes. Our technical meetings are riddled with nuclear physics, Monte Carlo calculations, cross sections, fluid dynamics, activities, radiation spectroscopy, and more. There are still dozens of isotope technologies waiting to be optimized by the right nuclear minds. More importantly, hospitals are continuously asking for more isotopes to do clinical research.
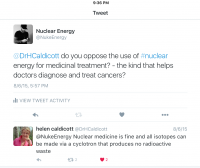 Oh, and the whole anti-nuclear thing? It's not as potent in nuclear medicine. Even the alleged nemesis of nuclear power, Dr. Helen Caldicott, is fine with medical isotopes based on her tweet.
Oh, and the whole anti-nuclear thing? It's not as potent in nuclear medicine. Even the alleged nemesis of nuclear power, Dr. Helen Caldicott, is fine with medical isotopes based on her tweet.
My dad had a procedure after a mild heart scare nearly 10 years ago. He was injected with the tagged isotope and asked to run on a treadmill to elevate his heart rate. This helped transport the isotope through his circulatory system and observe how his heart responded to stress. Where there was strong radiation emitting from my dad signaled healthy blood flow. Where there were darker spots, however, eluded to a weaker flow in that area. The whole procedure took several hours and the doctor immediately called afterward demanding that he go into surgery the next day, possibly saving his life.
When one thinks about the life cycle of my dad's isotope (Mo-99/Tc-99m)-the uranium mining, enrichment, fabrication, burnup, fission, purifications, gamma spectroscopy, and disposal-it should all sound familiar. That's nuclear science and engineering.
 Alex Brown earned a B.S. in chemical engineering, an M.S. in radiation physics, and a Ph.D. in radiochemistry. He is a member of the ANS Biology and Medicine Division, as well as the Fuel Cycle and Waste Management Division. He is a member of the Young Members Group. He lives in Chicago. You can follow him on Twitter.
Alex Brown earned a B.S. in chemical engineering, an M.S. in radiation physics, and a Ph.D. in radiochemistry. He is a member of the ANS Biology and Medicine Division, as well as the Fuel Cycle and Waste Management Division. He is a member of the Young Members Group. He lives in Chicago. You can follow him on Twitter.

